
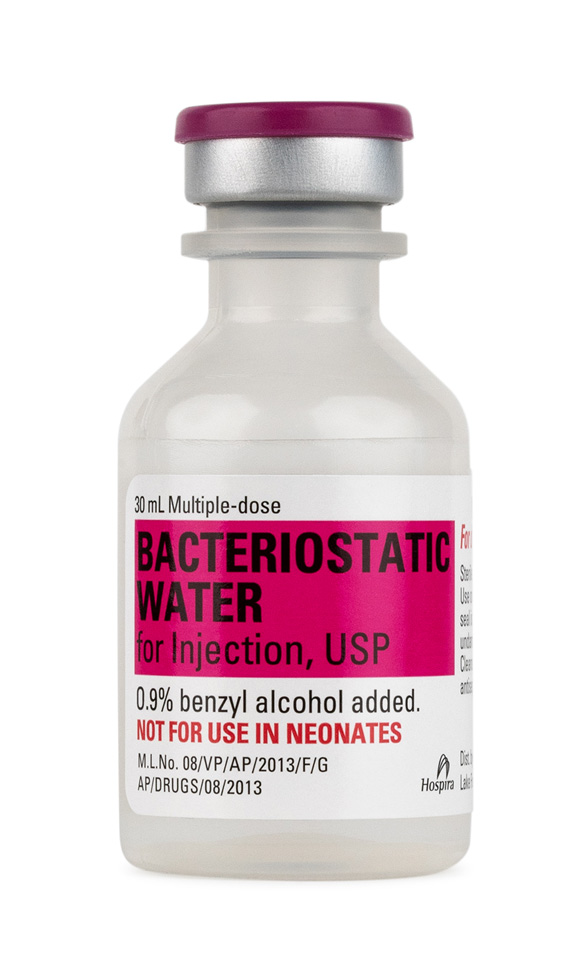
Hospira BAC Water
$35.00
BAC Water via Pfizer
Safety Data Sheet
1. Identification of the Substance/Mixture and the Company/Undertaking
Material Name: Bacteriostatic Water for Injection, USP (Hospira Inc.)
Trade Name: Bacteriostatic Water for Injection, USP
Chemical Family: Not determined
Intended Use: Pharmaceutical product
Details of the Supplier
Supplier (US): Hospira, A Pfizer Company | 275 North Field Drive | Lake Forest,
Illinois 60045 | Phone: 1-800-879-3477
Supplier (UK): Hospira UK Limited | Horizon, Honey Lane | Hurley, Maidenhead, SL6 6RJ
| United Kingdom
Emergency Telephone
CHEMTREC (24 hours): 1-800-424-9300
International CHEMTREC (24 hours): +1-703-527-3887
Contact Email: [email protected]
2. Hazards Identification
GHS Classification: Not classified as hazardous
Signal Word: Not Classified
Hazard Statements: Not classified in accordance with international standards for
workplace safety.
Other Hazards: No data available
Note: This document has been prepared in accordance with standards for workplace safety, which
requires the inclusion of all known hazards of the product or its ingredients regardless of the potential risk.
The precautionary statements and warnings included may not apply in all cases. Your needs may vary depending
upon the potential for exposure in your workplace.
3. Composition / Information on Ingredients
| Ingredient | CAS Number | EU EINECS/ELINCS List | GHS Classification | % |
|---|---|---|---|---|
| Benzyl Alcohol | 100-51-6 | 202-859-9 | Acute Tox. 4 (H302); Acute Tox. 4 (H332) | 1.1% |
| Water for Injection | 7732-18-5 | 231-791-2 | Not Listed | Balance |
Additional Information: Proprietary Ingredient(s) indicated as hazardous have been
assessed under standards for workplace safety. In accordance with 29 CFR 1910.1200, the exact percentage
composition of this mixture has been withheld as a trade secret.
For the full text of the CLP/GHS abbreviations mentioned in this Section, see Section 16.
4. First Aid Measures
Eye Contact: Flush eye(s) immediately with plenty of water. If irritation occurs or
persists, get medical attention.
Skin Contact: Rinse with plenty of water. If skin irritation persists, call a
physician.
Ingestion: Never give anything by mouth to an unconscious person. Wash out mouth with
water. Do not induce vomiting unless directed by medical personnel. Seek medical attention immediately.
Inhalation: Move to fresh air. If discomfort occurs, get medical attention.
Most Important Symptoms and Effects: For information on potential signs and symptoms
of exposure, See Section 2 – Hazards Identification and/or Section 11 – Toxicological Information.
Medical Conditions Aggravated by Exposure: None known
Notes to Physician: None
5. Fire Fighting Measures
Extinguishing Media: As for primary cause of fire.
Hazardous Combustion Products: Formation of toxic gases is possible during heating or
fire.
Fire/Explosion Hazards: Not applicable
Advice for Fire-Fighters: During all fire fighting activities, wear appropriate
protective equipment, including self-contained breathing apparatus.
6. Accidental Release Measures
Personal Precautions: Personnel involved in clean-up should wear appropriate personal
protective equipment (see Section 8). Minimize exposure.
Environmental Precautions: Place waste in an appropriately labeled, sealed container
for disposal. Care should be taken to avoid environmental release.
Methods for Containment and Cleaning: Contain the source of spill if it is safe to do
so. Collect spill with absorbent material. Clean spill area thoroughly.
Additional Consideration for Large Spills: Non-essential personnel should be
evacuated from affected area. Report emergency situations immediately. Clean up operations should only be
undertaken by trained personnel.
7. Handling and Storage
Precautions for Safe Handling: Avoid breathing vapor or mist. Avoid contact with
eyes, skin and clothing. When handling, use appropriate personal protective equipment (see Section 8). Wash
thoroughly after handling. Releases to the environment should be avoided. Review and implement appropriate
technical and procedural waste water and waste disposal measures to prevent occupational exposure or
environmental releases.
Storage Conditions: Store as directed by product packaging. Do not refrigerate.
Incompatible Materials: None known
Specific End Use: Pharmaceutical product
8. Exposure Controls / Personal Protection
Occupational Exposure Limits
| Region/Source | Limit Value |
|---|---|
| Pfizer OEL TWA-8 Hr | 10 ppm |
| Bulgaria OEL TWA | 5.0 mg/m³ |
| Czech Republic OEL TWA | 40 mg/m³ |
| Finland OEL TWA | 10 ppm / 45 mg/m³ |
| Latvia OEL TWA | 5 mg/m³ |
| Lithuania OEL TWA | 5 mg/m³ |
| Poland OEL TWA | 240 mg/m³ |
Engineering Controls: Engineering controls should be used as the primary means to
control exposures. General room ventilation is adequate unless the process generates dust, mist or fumes. Keep
airborne contamination levels below the exposure limits.
Personal Protective Equipment
- Hands: Wear impervious gloves (e.g. Nitrile, etc.) if skin contact is possible.
(Must meet EN374, ASTM F1001 or international equivalent.) - Eyes: Wear safety glasses as minimum protection. (Must meet EN166, ANSI Z87.1 or
international equivalent.) - Skin: Impervious protective clothing is recommended if skin contact with drug
product is possible and for bulk processing operations. (Must meet EN13982, ANSI 103 or international
equivalent.) - Respiratory: Under normal conditions of use, if the applicable OEL is exceeded,
wear an appropriate respirator with a protection factor sufficient to control exposures to below the OEL (e.g.
particulate respirator with a half mask, P3 filter). (Must meet EN140, EN143, ASTM F2704-10 or international
equivalent.)
9. Physical and Chemical Properties
| Physical State: Liquid | Color: Colourless |
| Odor: No data available | Odor Threshold: No data available |
| Molecular Formula: Mixture | Molecular Weight: Mixture |
| pH: 4.5–7.0 | Melting/Freezing Point: No data available |
| Boiling Point: No data available | Flash Point (Liquid): No data available |
| Evaporation Rate: No data available | Vapor Pressure: No data available |
| Vapor Density: No data available | Relative Density: No data available |
| Viscosity: No data available | Water Solubility: Soluble |
| Decomposition Temp: No data available | Autoignition Temp: No data available |
10. Stability and Reactivity
Reactivity: No data available
Chemical Stability: Stable under normal conditions of use.
Possibility of Hazardous Reactions: None
Oxidizing Properties: None known
Conditions to Avoid: None known
Incompatible Materials: None known
Hazardous Decomposition Products: None known
11. Toxicological Information
General Information: The information included in this section describes the potential
hazards of the individual ingredients. May cause eye irritation (based on components).
Acute Toxicity (Benzyl Alcohol)
| Species | Route | Result (LD50) |
|---|---|---|
| Rat | Oral | 1230 mg/kg |
| Mouse | Oral | 1360 mg/kg |
| Rabbit | Dermal | 2 g/kg |
Carcinogen Status: None of the components of this formulation are listed as a
carcinogen by IARC, NTP, or OSHA.
12. Ecological Information
Environmental Overview: Environmental properties have not been investigated. Releases
to the environment should be avoided.
Aquatic Toxicity (Benzyl Alcohol)
| Species | Test Type | Duration | Result |
|---|---|---|---|
| Fathead Minnow | NPDES LC-50 | 96 Hours | 460–770 mg/L |
| Bluegill | NPDES LC-50 | 96 Hours | 10 mg/L |
| Daphnia Magna (Water Flea) | Surrogate ErC50 | 48 Hours | 23–400 mg/L |
Persistence and Degradability: No data available
Bio-accumulative Potential: No data available
Mobility in Soil: No data available
13. Disposal Considerations
Waste Treatment Methods: Dispose of waste in accordance with all applicable laws and
regulations. Member State specific and Community specific provisions must be considered. Considering the
relevant known environmental and human health hazards of the material, review and implement appropriate
technical and procedural waste water and waste disposal measures to prevent occupational exposure and
environmental release. It is recommended that waste minimization be practiced. The best available technology
should be utilized to prevent environmental releases. This may include destructive techniques for waste and
wastewater.
14. Transport Information
The following refers to all modes of transportation:
- USDOT: Not regulated
- EU ADR: Not regulated
- IATA: Not regulated
- IMDG: Not regulated
15. Regulatory Information
Water for Injection
| CERCLA/SARA 313 Emission Reporting | Not Listed |
| California Proposition 65 | Not Listed |
| US TSCA Section 8(b) | Present |
| Australia (AICS) | Present |
| REACH Annex IV Exemptions | Present |
| EU EINECS/ELINCS List | 231-791-2 |
Benzyl Alcohol
| CERCLA/SARA 313 Emission Reporting | Not Listed |
| California Proposition 65 | Not Listed |
| US TSCA Section 8(b) | Present |
| Australia (AICS) | Present |
| EU EINECS/ELINCS List | 202-859-9 |
16. Other Information
Text of CLP/GHS Abbreviations:
- H302: Acute toxicity, oral – Cat. 4; Harmful if swallowed
- H332: Acute toxicity, inhalation – Cat. 4; Harmful if inhaled
Data Sources: Pfizer proprietary drug development information. Publicly available
toxicity information.
Revision Date: 25-Jul-2016
Prepared by: Pfizer Global Environment, Health, and Safety Operations — Product
Stewardship Hazard Communication
Disclaimer: Pfizer Inc believes that the information contained in this Material Safety Data
Sheet is accurate, and while it is provided in good faith, it is without warranty of any kind, expressed or
implied. If data for a hazard are not included in this document there is no known information at this time.
Copy

Reviews
There are no reviews yet.